Osteoporosis Treatment - #56
Take QuizPrimary and secondary options for therapy of osteoporosis.
Older adult in the outpatient setting
-
Identify indications for initiating osteoporosis treatment.
-
List interventions in reducing fracture risk.
-
List first line and second line osteoporosis therapy.
Treating primary osteoporosis
Osteoporosis is a silent disease that is often not identified until it is complicated by a fracture. Nearly 10 million Americans have osteoporosis and another 43 million people have low bone density.1 Approximately one out of every two Caucasian women and one in five men will experience an osteoporosis-related fracture in their lifetime.2
Eighty-six to 95% of low impact falls in persons with osteoporosis result in fractures. Hip fractures alone are associated with an 8-36% excess one-year mortality and 20% risk of long-term nursing home care.3,4 While hip fracture rates are greater in women than men, mortality from hip fractures is greatest in men. Hip fractures are also associated with a 2.5 fold increase in secondary fracture, making secondary prevention of osteoporosis and falls imperative to post fracture treatment plans.5
Science Principles
Treatment may be indicated even if a person does not meet clinical or radiological criteria for osteoporosis as the goal of treatment is to reduce the risk of fractures. Initiation of medical therapy is based on future fracture risk and not purely a radiographic finding (see Geriatric Fast Facts: Osteoporosis Screening and Diagnosis).
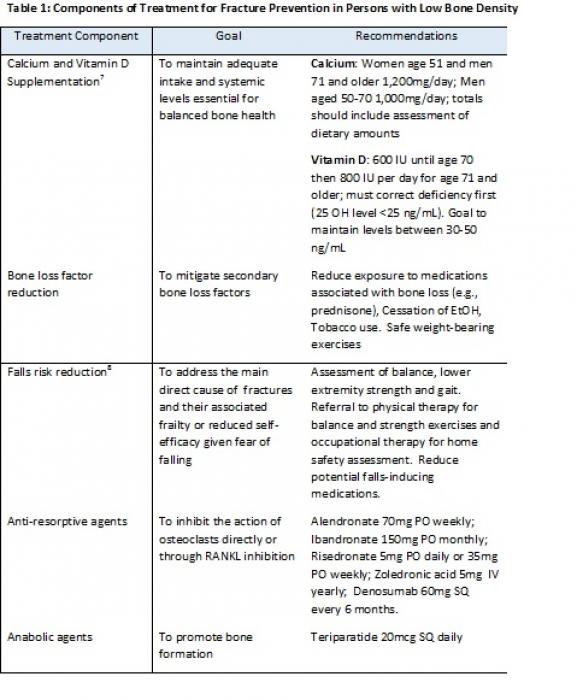
Treatment should include adequate calcium and vitamin D supplementation, mitigation of risk factors causing bone loss and reduction of fall risk (Table 1) as well as potentially other pharmacotherapy.
Indications for pharmacotherapy with a bisphosphonate, a RANKL (receptor activator of nuclear factor kappa-B ligand) inhibitor or an anabolic agent include:
-
Diagnosis of osteoporosis (as defined in Geriatric Fast Facts: Osteoporosis Screening and Diagnosis)
-
Osteopenia (T-score -1.0 to -2.4) and 10-year probability of hip fracture >3 percent or major osteoporosis-related fracture >20 percent based on the WHO FRAX calculator (https://www.shef.ac.uk/FRAX/tool.jsp).
Bisphosphonates are effective first line treatments for osteoporosis. They come in oral (alendronate, risedronate, and ibandronate) or intravenous formulations (zoledronic acid and ibandronate). Prior to initiating therapy with a bisphosphonate, it is imperative to check kidney function (requires GFR>35 mL/min/1.73 m2) and vitamin D status (level should be >25ng/mL to reduce the risk of hypocalcemia). A history of severe gastroparesis, esophagitis, stricture or other esophageal or stomach disorder may be relative contraindications to oral therapy. Side effects may include myalgias, fever, and fatigue, especially for IV preparations. Rare, but severe, side effects may include atypical fractures and osteonecrosis. Atypical fractures are rare and are associated with extended treatment (>10yrs) which is why a drug “holiday” (discontinuation) is introduced after 5 years of therapy. Osteonecrosis of the jaw is rare, with most cases occurring in cancer patients or in patients who were treated with high doses of IV bisphosphonates.6
Denosumab (a RANKL inhibitor) is not considered as initial therapy for most patients with osteoporosis though it may be appropriate for patients at high risk for fracture who have a contraindication to, or unresponsiveness towards, bisphosphonates, or those patients whose GFR < 35mL/min/1.73m2. This medication is administered every 6 months by subcutaneous injection. The side effects are similar to bisphosphonates and include the risk of atypical fractures and osteosarcoma. Risk of infections, including skin, abdominal, lung, and urinary system are slightly more common (4%) and if developed while on therapy are a reason for discontinuation of any future treatments.
Teriparatide is an anabolic agent stimulating bone formation through activation of bone remodeling. This medication is not considered initial therapy for most patients and is reserved for those who have severe osteoporosis (T-score of -3.5 or below even in the absence of fractures, or T-score of -2.5 or below plus a fragility fracture), have a contraindication to bisphosphonates, or failed other osteoporosis therapies. Treatment requires daily subcutaneous injections. Side effects may include bone pain, injection site irritation, and hypercalcemia. Rare risks include osteosarcoma. Treatment duration is two years.
Review of Systems (ROS)
Geriatric Topics
ACGME Compentencies
Science Principles
Users are free to download and distribute Geriatric Fast Facts for informational, educational and research purposes only. See Term of Use for additional information.
Disclaimer: Geriatric Fast Facts are for informational, educational and research purposes only. Geriatric Fast Facts are not, nor are they intended to be, medical advice. Health care providers should exercise their own independent clinical judgment when diagnosing and treating patients. Some Geriatric Fast Facts cite the use of a product in a dosage, for an indication, or in a manner other than that recommended in the product labeling. Accordingly, the official prescribing information should be consulted before any such product is used.
Terms of Use: Geriatric Fast Facts are provided for informational, educational and research purposes only. Use of the material for any other purpose constitutes infringement of the copyright and intellectual property rights owned by the specific authors and/or their affiliated institutions listed on each Fast Fact. By using any of this material, you assume all risks of copyright infringement and related liability. Geriatric Fast Facts may not be reproduced or used for unauthorized purposes without prior written permission, which may be obtained by submitting a written request to: Medical College of Wisconsin, Dept. of Medicine, Division of Geriatrics and Gerontology, 8701 Watertown Plank Road, Milwaukee, WI 53226. Note the Geriatric Fast Facts may contain copyrighted work created under contract with government agencies, foundations, funding organizations and commercial companies, etc. If a particular author places further restrictions on the material, you must honor those restrictions regardless of whether such restrictions are described in this mobile app.